US patient receives first CSIRO-Foldax designed Tria polymer heart implant

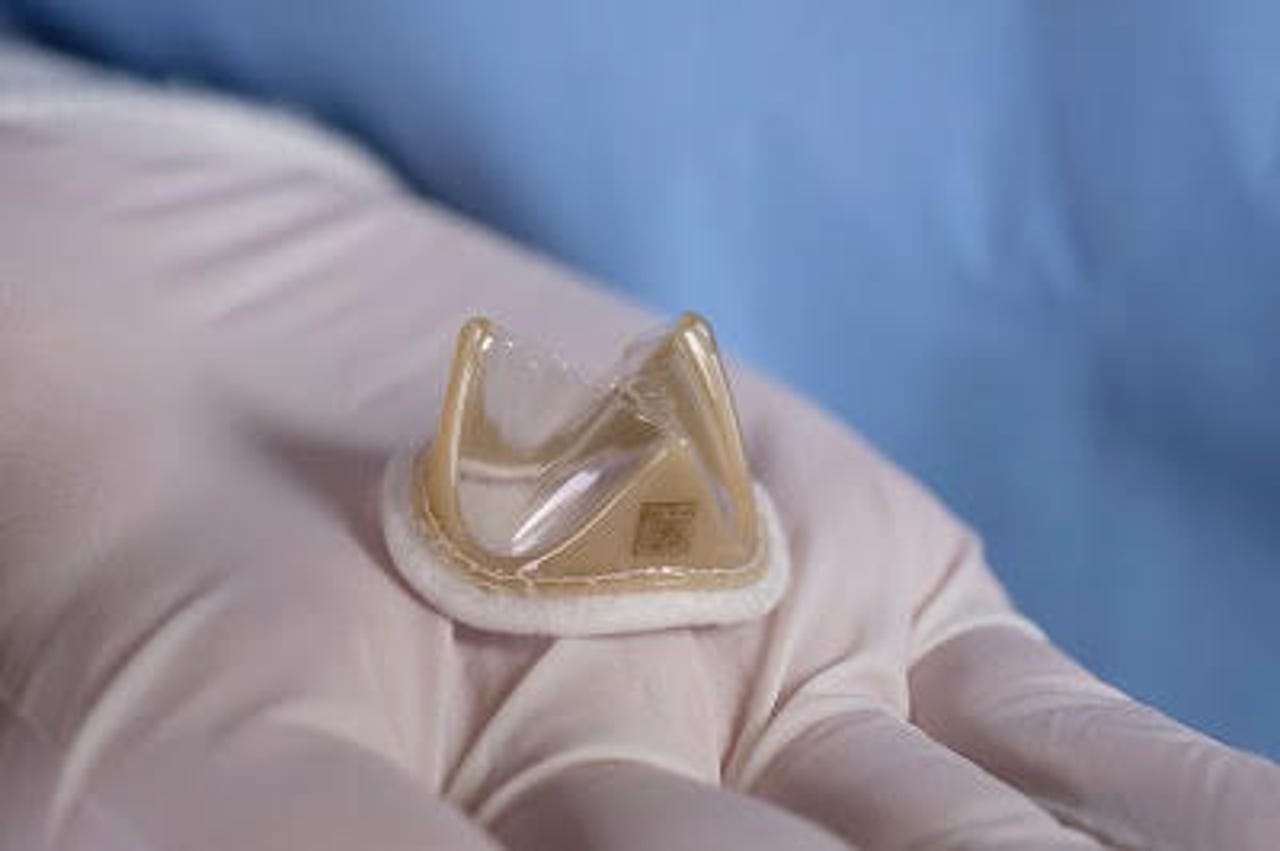
A patient at Beaumont Hospital in Michigan is in recovery after receiving a polymer heart valve implant in July.
Jointly developed by the Commonwealth Scientific and Industrial Research Organisation (CSIRO) and US-based medical device company Foldax, the Tria heart valve is made from CSIRO's proprietary polymer technology -- the same organisation that developed extended wear contact lenses and biomedical polymers such as those found in cardiac pacemakers.
According to CSIRO, using polymer for the Tria heart valve creates a valve that is capable of lasting decades without calcification, risk of clotting, or damage to red blood cells.
"This is a true example of Aussie innovation going global, with our team in Melbourne designing, developing, and scaling-up the new polymer," CSIRO project leader Dr Thilak Gunatillake said.
See also: Google uses AI, deep learning to predict cardiovascular risk from retina scans (TechRepublic)
Foldax now manufactures the Tria heart valve for patients from its headquarters in Salt Lake City, Utah.
"Tria heart valves are revolutionising the industry as the first and only biopolymer heart valve platform using LifePolymer material, eliminating the use of animal tissue," said Foldax executive chairman Ken Charhut.
"What makes this so different from other heart valves is that we were able to design the valve to mimic the native valve."
The CSIRO said it is in discussions with potential industry and research partners to translate the technology into further applications, such as vascular grafts or synthetic membranes for repair of eardrum ruptures.
The operation is the latest success story for the CSIRO, which previously teamed up with Melbourne-based medical implant company Anatomics and doctors in the United Kingdom to implant a 3D-printed titanium and polymer sternum into a 61-year-old British patient.
The 3D-printed sternum was designed by Anatomics and printed at CSIRO's Lab 22 facility in Melbourne. It was the first time a titanium sternum combined with a synthetic polymer had been used to replace bone, cartilage, and tissue in a patient.
In 2015, the CSIRO-Anatomics partnership saw a 54-year-old cancer patient from Spain receive a 3D-printed titanium sternum and rib implant that was designed and manufactured out of Lab22.
Updated 30 September 2019, 3.30pm AEST: The polymer used for the heart valve is different to the polymer used for Australia's bank notes, extended wear contact lenses, and other biomedical polymers.
Related coverage
CSIRO's Data61 develops 'vaccine' against attacks on machine learning
Data61 claims its new set of vaccination-like techniques show it's possible to prevent adversarial attacks made on machine learning algorithms.
Data61 using machine learning to track human infectious diseases in Australia
The tool uses statistical machine learning to hopefully prevent the spread of infectious diseases in Australia.
CSIRO's Data61 opens Mixed Reality Lab in Melbourne
The lab will allow industries to create digital copies of physical objects, systems, or people.
CSIRO using serverless compute to analyse the human genome
The CSIRO is using AWS Lamba to allow analysis of the 20 exabytes of data coming from genomics every year.