EmTech MIT: How AI and biomedical innovations are transforming health care

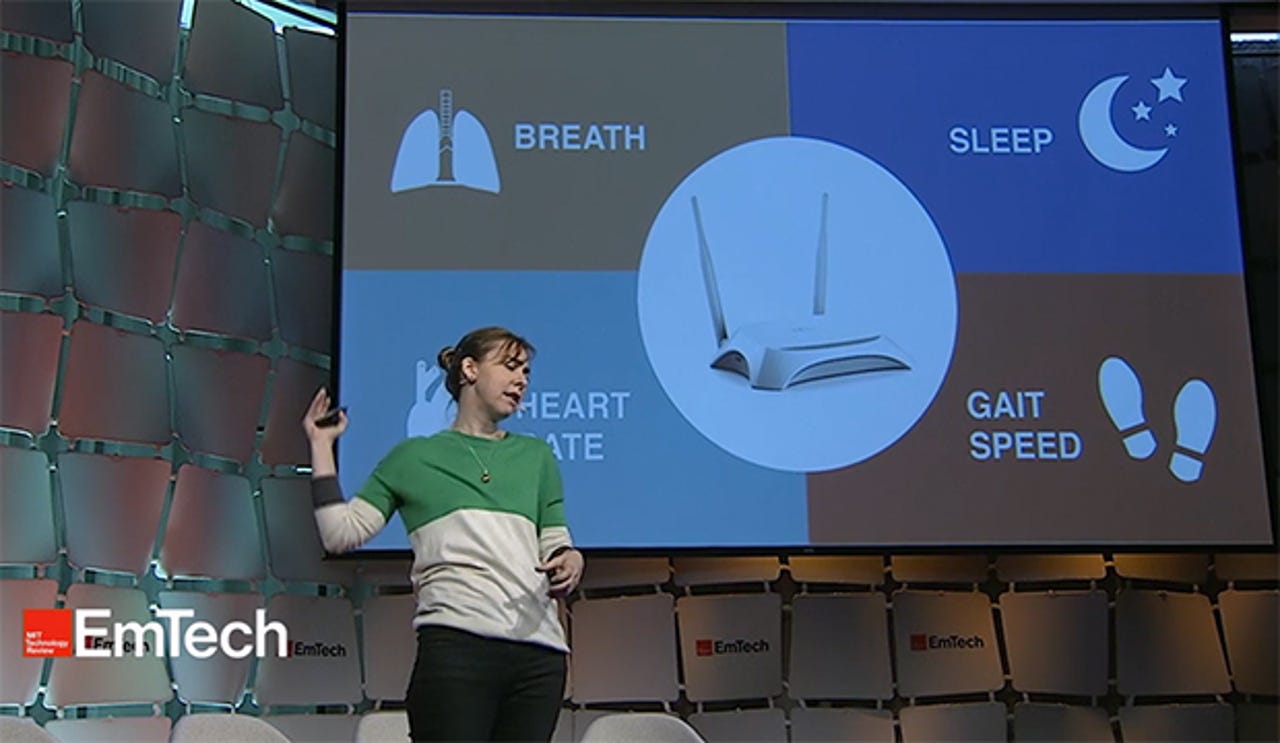
The way that doctors monitor breathing, detect falls, track movement and gait in patients with Parkinson's or MS or analyze sleep requires invasive sensors and often needs to be done in a specialized setting. Other signs are simply too difficult to monitor so patients must write in a diary instead.
MIT professor Dina Katabi's group has developed a prototype wireless device much like a WiFi router that uses a combination of radio signals and machine learning algorithms to monitor these physiological signs without wires--even through walls. In a presentation at last week's EmTech MIT conference, Katabi described how the system works and some of the potential applications.
"We are all swimming in a sea of wireless signals, and every movement changes that electromagnetic field" because our bodies are primarily made of water and reflect these signals, Katabi said. The device sits in the background and analyzes these changes in the electromagnetic field using advanced machine learning algorithms to extract physiological data. Katabi claimed the system can measure breathing, heart rate, gait, sleep and a variety of other physiological signals throughout a home.
In a video demonstration, Katabi showed how the system, dubbed Emerald, can accurately track movement, detect a fall, and monitor heart rate with an accuracy of about 97 percent in comparison with an FDA-approved chest band, even for multiple occupants in a home. It can not only monitor sleep, but also distinguish between the different stages of sleep and diagnose sleep apnea, which can be indicative of disorders such as depression or Alzheimer's.
Emerald is currently being tested in about 200 homes with both healthy individuals and patients suffering from Alzheimer's, depression, Parkinson's, and pulmonary disease. By tracking the trajectory of people in assisted-living facilities over time, it can generate data that provides lots of insights about mobility, the efficacy of medications, and overall mental and physical health.
The ultimate goal, Katabi said, is a "health-aware home" that is sits in the background and constantly keeps tabs on us for the doctor. "In the future, the health care should come to the patient in their homes," Katabi said. "In fact, they should hardly ever need to go to a clinic or hospital."
Emerald was just one of several talks last week on innovations in health care.
Sheng Xu, an assistant professor in the Department of Nanoengineering at UC San Diego, described another approach to less-invasive health monitoring. Stretchable electronics can capture a variety of physical and chemical signals, and send them wirelessly to a cell phone or the cloud, but these are literally skin deep. The Xu Research Group developed a stretchable ultrasound device that can see through the skin to capture more vital signs. For example, the device can be worn on the neck to monitor the carotid artery and jugular vein well below the skin's surface.
Frequency Therapeutics has just started the first clinical trial of a so-called Progenitor Cell Activation (PCA) platform, a proprietary compound of small molecules that activates the inner ear's cells to regenerate, or divide and form new sensory cells, potentially reversing hearing loss. Co-founder and vice president Will McLean said that the human body has some tissues that naturally regenerate, but nothing like the intestine, which completely regenerates its lining every five to seven days. Frequency Therapeutics developed small-molecule drugs that can control this process in the intestine. The cochlea happens to be a close cousin--it shares the same progenitor cell but it is not regenerative--so by using the same drugs, Frequency Therapeutics hopes to reverse hearing loss. Eventually, McLean hopes, the same techniques could be applied to other tissues such as the brain, eyes, muscles, cartilage, skin and hair,
James Dahlman, an assistant professor at the Georgia Institute of Technology, talked about the use of nanoparticles to deliver drugs to the right tissue. This "sounds like science fiction," Dahlman said, but last month the FDA approved the first genetic therapy that uses nanoparticles to treat liver disease. The catch is that we can still only design drugs that deliver medicine to the liver and the current method for testing new nanoparticles isn't scalable. Dahlman's lab figured out a way to test thousands of nanoparticles at once in a single mouse using DNA barcodes and gene sequencing to track where everything went. They are hoping this technique will lead to the discovery of safer and more effective drug treatments.
Shinjini Kundu, a physician and medical researcher at University of Pittsburgh Medical Center, developed a medical imaging technique called transport-based morphometry (TBM) that uses AI to spot latent diseases imperceptible to humans reviewing MRIs. In a study of 70 subjects, Kundu's system was able to predict signs of osteoarthritis in healthy people up to three years before symptoms became visible with 86 percent accuracy.
Adam Marblestone, the Chief Strategy Officer at Kernel, talked about bleeding-edge efforts to develop new ways to record what is going on in the brain. The current approaches using invasive electrodes or MRIs aren't scalable, he said. We can record only one thousand neurons and at the current rate of progress it would take until the end of the century just to record all the neurons in a mouse brain--let alone the 100 billion neurons in the human brain. Instead, Marblestone said, we need to completely rethink the physical interface to the brain--an effort that began with the Obama administration's Brain Initiative.
One interesting idea is to re-engineer the cells in the brain to act like a hard drive and record their own activity in a way that could be read out later. Those experiments are fine for researchers working with animals, but they aren't suitable for humans for obvious reasons. Last year Marblestone joined Kernel, a $100 million startup backed by Bryan Johnson, founder of online payments company Braintree, that aims to develop a less invasive physical interface for neural recording to "accelerate our understanding of how the brain works and fix it when it doesn't."
Niki Bayat, is the co-founder and Chief Science Officer at AesculaTech, which has developed a smart material for medical devices that is a liquid at lower temperatures, turns into a gel when heated and becomes solid at body temperature. The gel is injectable, easily removable, and adapts to body shape. It can be inserted into the tear duct to release glaucoma medicine over an extended period. Bayat said that the company recently completed its first human trial showing that this is safe and effective treatment for glaucoma.