On the trail of counterfeit drugs

Consumers expect the medicines they buy to be authentic, but it is possible that they are not. Good news is there are technologies that can effectively thwart counterfeiting in pharmaceutical manufacturing.
According to Peter Stokes, regional director of e-trade at Axway Asia-Pacific, the World Health Organization (WHO) estimates that up to 50 percent of drugs worldwide are counterfeit. This translates to approximately US$38 billion in legitimate corporate sales lost each year due to the sale of counterfeit drugs.
The Center for Medicines in the Public Interest also predicts that counterfeit drug sales could reach US$75 billion globally in 2010, if action is not taken to curb the illegal trade, Stokes told ZDNet Asia in an e-mail.
He said counterfeit drugs make their way through the supply chain via various means, one of which is the parallel import of pharmaceuticals. This, he noted, potentially exposes a product to tampering and patients to counterfeits.
Counterfeiters have also taken advantage of the many different channels that drugs are processed, each at different price points. In channel diversion, for instance, an unauthorized reseller can move a product from a low-cost channel into a high-cost category. Similar to issues faced by parallel trade, channel diversion creates patient safety risks from expired products or tampered packaging being sold through Internet channels at discounts to vulnerable buyers, Stokes said.
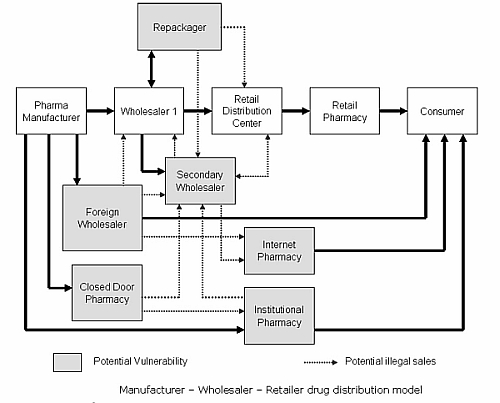
Kozhikote Jayan, director of pharma and life sciences at Hewlett-Packard Asia-Pacific, said the counterfeit drug trade puts everyone in the supply chain at risk, ranging from raw material providers to manufacturers, distributors, wholesalers, logistics service providers, pharmacies and distribution institutions, such as clinics and hospitals.
"Patients are at risk as they do not receive the intended benefits of the real drug," Jayan said in an e-mail interview. "The quality of material, manufacturing process and counterfeit packaging affects the safety of a drug."
Stokes said IT plays an important role in the fight against counterfeiting, from the automation of processes to data monitoring, management of serialized numbers, streamlining of data exchange and business processes, and helps ensure security along the entire supply chain.
Stamping out the problem
There are two main methods today to combat drug counterfeiting: "pedigree" and "track and trace".
A drug pedigree, Stokes explained, is a statement of origin that identifies each prior sale, purchase or trade of a drug. It includes the date of transactions, as well as names and addresses of all parties involved in the transactions. As a drug moves from the manufacturer through the supply chain, each company along the process adds to the pedigree.
The seller identifies the drugs and the full chain of custody, certifies the pedigree of each and transmits this in advance. The trading partner receiving the drugs then authenticates the pedigree.
When the shipment arrives, the pedigree of the drug is matched to the product and signed, verifying its accuracy. The pedigree software maintains the product-to-pedigree match while the products are in inventory. The distributor can then send individual units to pharmacy customers, with each unit having an individual pedigree, Stoke said.
As paper pedigrees can be fairly easy to counterfeit, manually tedious and cost-prohibitive, more pedigrees are now being provided by electronic documents called ePedigrees. These must contain a digital signature created via only government-approved technology and verified by an automated system. The digital signature must also follow common PKI (public key infrastructure) practices, including the issuance and management of digital certificates.
The track-and-trace process keeps tabs on the serial numbers, product master data and distribution channel information over the product's lifecycle, Stokes said. Serialized and non-serialized events are managed along with the master data in an open framework to allow internal and external trusted parties access to product identification, status, location and event data at any point in the supply chain.
Serialized tracking and tracing radio frequency identification (RFID) and 2D barcode technology are used to identify an individual bottle of drugs, and trace its entire lifecycle from manufacturing to wholesaling, distribution, retailing and dispensing.
According to Stokes, Axway deploys both types of security processes to ensure the safety of the US$1.3 billion in healthcare transactions a day, that go through the company's business-to-business infrastructure.
RFID's the way
HP's Jayan said deploying RFID and global positioning system (GPS) technologies along the drug supply chain helps to combat counterfeiting. "Ultimately, RFID tags make business sense because they help in tracking the consignment at every stage."
Lum Seow Khun, public sector country manager at IBM Singapore, said RFID fulfills the criteria that anti-counterfeiting technology must be cost-effective, adaptable and scalable.
The capacity of RFID tags to contain enough data for the identification of unique items, combined with their ability to layer encryption and additional features, lets companies raise operating efficiencies and decrease labor costs, while simultaneously increasing security levels, said Lum in an e-mail.