Should we all be guinea pigs?

"Objects in mirror are closer than they appear" is the familiar warning inscribed on cars' rear view mirrors, but the same could be said for "technologies in the future." We can grow up dreaming of amazing inventions off on the horizon of tomorrow, and we may watch as they get closer year by year. Even so, we can still be stunned to realize suddenly that they are nearly upon us, and can maybe even feel a moment's anxiety that despite all the advance notice, we're not quite ready for them.
Recently, I had two conversations in one day that brought home how imminent certain advances could be in the lives of people I care about. Regenerative medicine is becoming less exotic by the day, and medical tourism is opening up access to it for those willing to cut some corners by crossing international borders. Meanwhile, the ongoing digitization of recordkeeping could lead to the disappearance of all boundaries, including some that may have traditionally helped to protect medical consumers from quacks -- and themselves.
First conversation: stem cells and scleroderma
Tuesday morning three weeks ago, my friend Bob called me, looking for my opinion and whatever science information I might be able to offer. I wasn't surprised that the subject was his wife Alice, who was diagnosed a few months ago with the condition called scleroderma. (For the sake of their privacy, I have changed the names of all the people and certain other details in this story.)
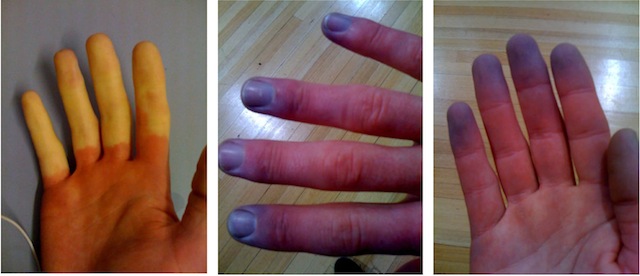
Scleroderma is a poorly understood illness that is generally classified as a chronic autoimmune disorder. The name, which means "hardened skin," describes one of its most obvious consequences. Small blood vessels, particularly in the skin, start to die and they are replaced inappropriately with the fibrous connective protein collagen. Patients can start to develop calcified patches on their skin, or the skin itself can become tight and hard. The severity of scleroderma can vary considerably. If the skin hardening becomes severe enough, movement of some joints may become painful or impossible. In more severe systemic cases, internal organs may be affected, which can become lethal.
Unfortunately, no truly satisfactory treatments exist. Some patients find that corticosteroids or other immune suppressors can slow the progress of the disease, but many therapies simply concentrate on decreasing the pain or poor circulation that patients feel.
Alice has been lucky in that, so far, the scleroderma seems to have been restricted to her skin. She may have been living with the preliminary stages of scleroderma for many years: like many patients, she was diagnosed years earlier with Raynaud's syndrome, in which circulation to the fingers shows exaggerated reactions to cold and heat.
Nevertheless, Alice was becoming extremely uncomfortable, and both she and Bob feared how the continued progression might change her for the worse. After the standard options for treatment proved frustrating, Alice and Bob had started investigating less orthodox ones.
And that was why, Bob told me, they had signed up Alice for experimental stem cell treatments. In China.
"What do you think?" Bob asked.
Medical tourism isn't science
My first thought was that to call the treatment experimental seemed almost too generous. Immensely promising as they are in the nascent field of regenerative medicine, stem cells have already been put under a cloud by controversial clinics in various countries that make dubious claims about treating illnesses like cancer that often confound conventional cures.
A quick check of the published literature showed that Daniel E. Furst of UCLA's School of Medicine and other researchers had indeed conducted pilot clinical studies on treating scleroderma with stem cells. The concept was that if the body's immune system had turned against it, then perhaps a "reboot" of immunity with fresh cells might alleviate the disease.
Typically, the investigators had first used chemotherapy to kill off the patient's defective immune system, and then infused stem cells previously derived from the patient's own body -- starting either with isolated blood stem cells that had then been cultivated in the lab or with other body cells induced into a stem-like pluripotent state.
But in some experiments, doctors had tried using stem cells from donated umbilical cords rather than from the patients themselves. Some of them, such as Richard K. Burt of the Northwester University School of Medicine had also tried not giving chemo before administering the stem cells, in the hope that the presence of the stem cells might either curb the immune system's destructive side or support the survival of healthy, unafflicted skin.
Reassuring as it was to find some precedents in the literature, however, what Alice was agreeing to undergo didn't really seem to qualify as a proper scientific study at all, as far as I could tell from the incomplete information Bob was giving me. That she knew she would be getting the cells already suggested it wasn't anything like a double blind study. It also wasn't clear how many other patients might be getting these treatments, and whether the clinic was controlling for age, ethnicity, life history, or any of the other factors that could affect the outcomes of the disease and this treatment.
I gathered that Alice's therapy would involve cord stem cells and no chemotherapeutic agents. Her treatment could therefore be relatively quick and easy. Those facts certainly helped to explain why this course of action was so appealing to Alice -- but it also was exactly the kind of "gain with no pain" promised by many quack treatments. It seemed like what she was about to do had less in common with scientific study than with simple medical tourism.
My natural first impulse was to say that I thought it was a bad idea and that she shouldn't go. Bob, who had been studying up about all this and understood about the risks, was quick to agree with me that it was very much "voodoo medicine," in his words.
But it also became immediately apparent that the treatment was a fait accompli. Alice and Bob were both desperate to bring her some relief before her quality of life sank further, and they were determined to go ahead with this despite the risks. She was, in fact, already booked to leave on Friday and would return a week later.
I consoled myself with the knowledge that in the published medical studies of using stem cells against scleroderma, serious side effects had not yet been seen and about 50 percent of the patients had sometimes reported getting some relief for at least two years.
I was also glad to hear that, aside from the travel and accommodation costs, Alice and Bob weren't paying for the treatments (not this first time, anyway).
And even though I have often argued strenuously against medical quacks who take advantage of the desperately sick by pushing unproved treatments, I had to admit that under the circumstances I could sympathize enough with Alice and Bob to understand why they had made the choice that they did. I wished them the best.
(An interim update, if you will: Alice has been to China, had her treatments, and returned. So far she has not noticed any obvious improvements in her condition but she recognizes that it is still very early, and she has not experienced any unwanted side effects aside from some general itchiness. We'll see what happens.)
Second conversation: experiment on everyone
Later that evening after work, I met up for a drink with some colleagues who were passing through New York. One of them, Patricia, has a long background in the biotech industry and strong opinions about modernizing medicine and expediting FDA approval of new therapies.
Because Alice's situation and her choice of therapy were on my mind, I mentioned them to Patricia. Her response was not quite what I expected. She seemed to breeze past some of my concerns about the uncertainty of the treatment and instead saluted the idea of patients routing their way around what she saw as roadblocks to speedier progress.
"But part of what bothers me," I said, "is that whatever benefit Alice does or doesn't get out this treatment, it seems like the loose way in which it's being done isn't going to be much good for figuring out how to help other people."
"Sure," Patricia said, "but that's just because we don't have everybody enrolled in trials all the time. Once we fix the country's medical records problem, that won't matter anymore."
I knew what she was getting at. It's part of the promise of electronic medical records (EMRs).
There's been a lot of talk in recent years about getting the medical establishment to use standardized, digital recordkeeping more extensively. Among the benefits commonly touted are that EMRs could help to reduce medical errors, reduce recordkeeping costs, and guarantee to patients that their medical files that could follow them throughout life.
But in principle, EMRs could also revolutionize medical studies. Currently, finding patients who are willing to enroll in clinical trials is one of the major obstacles to research. Researchers don't even have as much information as they might like about ostensibly healthy people. Furthermore -- apropos Patricia's own passions -- it might be possible to get drugs approved more quickly if any elusive unwanted effects they had could be identified quickly in the general population.
EMRs -- and highly permissive rules about how they could be used -- could go a long way toward making that possible. If everybody's records could be appropriately anonymized and made accessible, many types of medical studies could be conducted retroactively by sifting through thousands or millions of relevant patients' health histories.
One such study was published earlier this year, for example, by R. H. Perlis and colleagues at Massachusetts General Hospital, who used EMRs to do a large-scale retrospective study in psychiatry.
Bringing EMRs to that level of usefulness more generally will be a major challenge, however -- procedurally, technologically, and legally. Physicians are often very attached to their own ways of keeping records, and they may not use precisely the same terminology even when while working in different departments of a single hospital. Creating a common EMR platform that they will all accept will take work, as will making sure that the EMRs can be accessed and searched efficiently from anywhere. And protecting the privacy of patients (and their physicians) within such a system will be essential.
It would also mean that people like Alice might be able to find the treatments that would best help them more easily, and that their experience might inform medical science even without their having enrolled in a clinical trial. And to Patricia's point, some patients might also find the freedom to pursue a wider range of medical options.
Reservations
The idea of using EMRs to improve the state of medicine and help biomedical research, and give people like Alice more options is undeniably appealing. Yet I would worry if one consequence of the system that resulted were that desperate people like my friend found too much encouragement (or insufficient discouragement) to pursue dubious treatments.
It isn't easy to stand against patients' freedom, but there's nothing to respect about medical practitioners who, either out of ignorance or callous greed, promote hollow remedies that are unlikely to deliver the sought benefits. Being on guard against the unscrupulous and incompetent and protecting sick people from their own frantic impulses are still necessary functions.
I'll very much hope that Alice's stem cell adventure ends well, and that the clinic in China she has visited will turn out to be the future global mecca for such scleroderma treatments. But at the moment, the therapy it offers seems too untested. If I had the opportunity to warn someone else away from it, I would.
This post was originally published on Smartplanet.com