After Samsung's Note 7 fires, researchers build mini extinguisher into battery

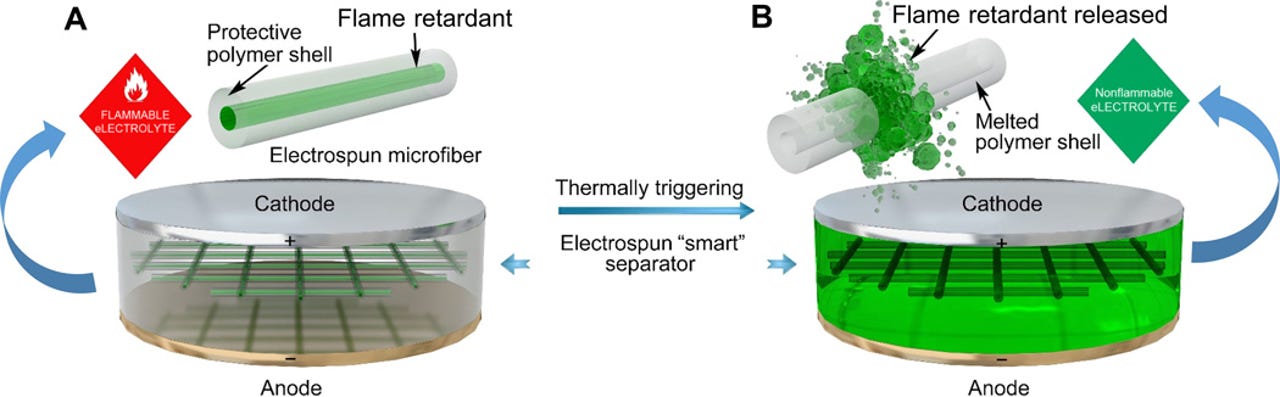
When triggered by high temperature, the polymer shell melts and releases the flame retardant into the electrolyte.
Scientists have developed a self-extinguishing lithium-ion battery designed to rapidly douse a fire caused by overheating.
Researchers from Stanford University report in a new paper published on Science Advances their use of a fire-retardant called triphenyl phosphate (TPP) contained within a polymer shell.
The shell sits inside a lithium-ion battery, shielding the battery's electrolytes from the retardant unless the battery reaches a certain temperature, at which point the polymer would melt and release the retardant.
The technology is aimed at higher-density lithium-ion batteries for electric vehicles, though the same risks have been known to affect batteries for personal devices. A report yesterday claimed Samsung's investigation into the Galaxy Note 7 has blamed its batteries for causing the phones to catch alight and explode.
The researchers argue their creation could open up new doors for safer, high-density applications for batteries.
"Although the energy densities of batteries continue to increase, safety problems remain a big issue, significantly hindering their further practical applications," the researches write.
"In the case of internal or external short circuits, undesirable exothermic reactions may lead to a rapid rise in internal temperature and to thermal runaway. The flammable liquid electrolytes would be ignited, eventually leading to fire and battery explosion," they explain.
They detail numerous other methods that have been used to address fire risks in lithium-ion batteries, but note drawbacks in all the techniques, which either sacrifice battery performance or only partially address fire risks.
The researchers describe both the fire retardant and the shell, which is made of poly(vinylidene fluoride-hexafluoropropylene), as a new answer to the risk.
The shell itself doesn't hinder battery performance since it prevents the retardants from mixing with the battery's electrolyte.
Also, since the retardant is shot directly into the electrolyte it can extinguish a fire more quickly. Tests showed the flames of the electrolyte were completely extinguished within 0.4 seconds.
The specific polymer also has a melting point of around 160°C (320°F), enabling it to release the retardant earlier in the combustion process, the researchers note.