Development boosts lithium-ion battery power by 8-fold

Lithium-ion batteries are the most common type of rechargeable battery. They are found in laptops, smartphones, and increasingly, in electric cars and smart grids.
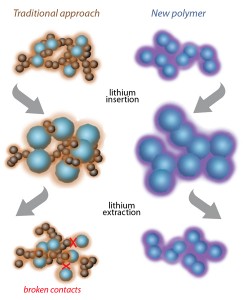
A team of scientists at Berkeley Lab have designed a new kind of anode that can absorb eight times the lithium of current designs, and has maintained its greatly increased energy capacity after over a year of testing and many hundreds of charge-discharge cycles.
“Most of today’s lithium-ion batteries have anodes made of graphite, which is electrically conducting and expands only modestly when housing the ions between its graphene layers. Silicon can store 10 times more – it has by far the highest capacity among lithium-ion storage materials – but it swells to more than three times its volume when fully charged, ” said Gao Liu of Berkeley Lab’s Environmental Energy Technologies Division (EETD).
The swelling quickly breaks the electrical contacts in the anode, so the researchers concentrated on finding other ways to use silicon while maintaining anode conductivity. Through a combination of synthesis, spectroscopy and simulation, the team tailored a polymer that conducts electricity and binds closely to lithium-storing silicon particles, even as they expand to more than three times their volume during charging and then shrink again during discharge.
The new anodes are made from low-cost materials, compatible with standard lithium-battery manufacturing technologies.
The research team reports its findings in Advanced Materials, now available online.
Source: Berkeley Lab News Center